🌍Curaleaf enters Europe through EMMAC acquisition ($286M)
EMMAC provides Curaleaf with a presence in key European Medical Cannabis Markets (U.K., Germany, Italy, Spain, and Portugal)
Press Release
Summary/Thoughts:
Transaction Value: $286M (85% stock — 16.8M shares @ $14.12 on 3/8/21; 15% cash — $50M); additional $57M (85% stock/15% cash) earnout (up to $343M)
EMMAC shareholders will own ~3% of Curaleaf before factoring in earn-outs
Assuming $40M Revenue based on management comments, Curaleaf paid ~7.1x Revenue
Brings Curaleaf into competition with Canadian LPs (Aphria/Tilray, Aurora, Canopy) who already have European operations, and Canndoc (go-public via Subversive SPAC announced in January)
EMMAC should contribute $40M in Revenue in 2021E and $100M in 2022E
Curaleaf is the first US MSO to meaningfully enter the European market — which I’m not sure is a good thing given that the company hasn’t absolutely solidified the #1 position in the U.S. (still a lot of whitespace). Management points out that this transaction is similar to buying into the footprint equal to Curaleaf’s U.S. presence back in 2017 ($19M), at a similar valuation. But EMMAC has twice the revenue of Curaleaf at that time, and has a larger potential addressable market than the U.S.
Measure 8 Ventures, an investment fund managed by Boris Jordan, Executive Chairman of the board of directors and control person of Curaleaf, has an interest in the proposed transaction by way of a profit interest and a convertible debt instrument which will convert into shares of EMMAC representing 8% of EMMAC equity at closing of the proposed transaction
Expect more European M&A from Curaleaf — Boris Jordan said he expects to make additional acquisitions in Europe, and getting into that market is a way to eventually expand into the Middle East and Africa
“We’ll be very acquisitive, similar to what we did in the U.S. Countries like Poland, Ukraine, South Africa — there are even rumors of Egypt — are also moving toward legalization and Europe is a great hub to attack those markets from. Our whole idea is to spread the Select brand around the word. We want to be the No. 1 brand.”
Increases the company’s TAM ➡️ ($100B U.S. + $229B Europe)
*Europe estimate derived from $100B U.S. estimate, adjusted for population ($100B USA market implies $305.81 annual spend per capita, $305.81 annual spend per capita x Europe population of 748M = $229B)
💭This probably overstates the TAM as disposable income is lower in Europe. Brightfield Group estimates the market at $8B by 2023. Arcview estimates it at $3.8B by 2025.
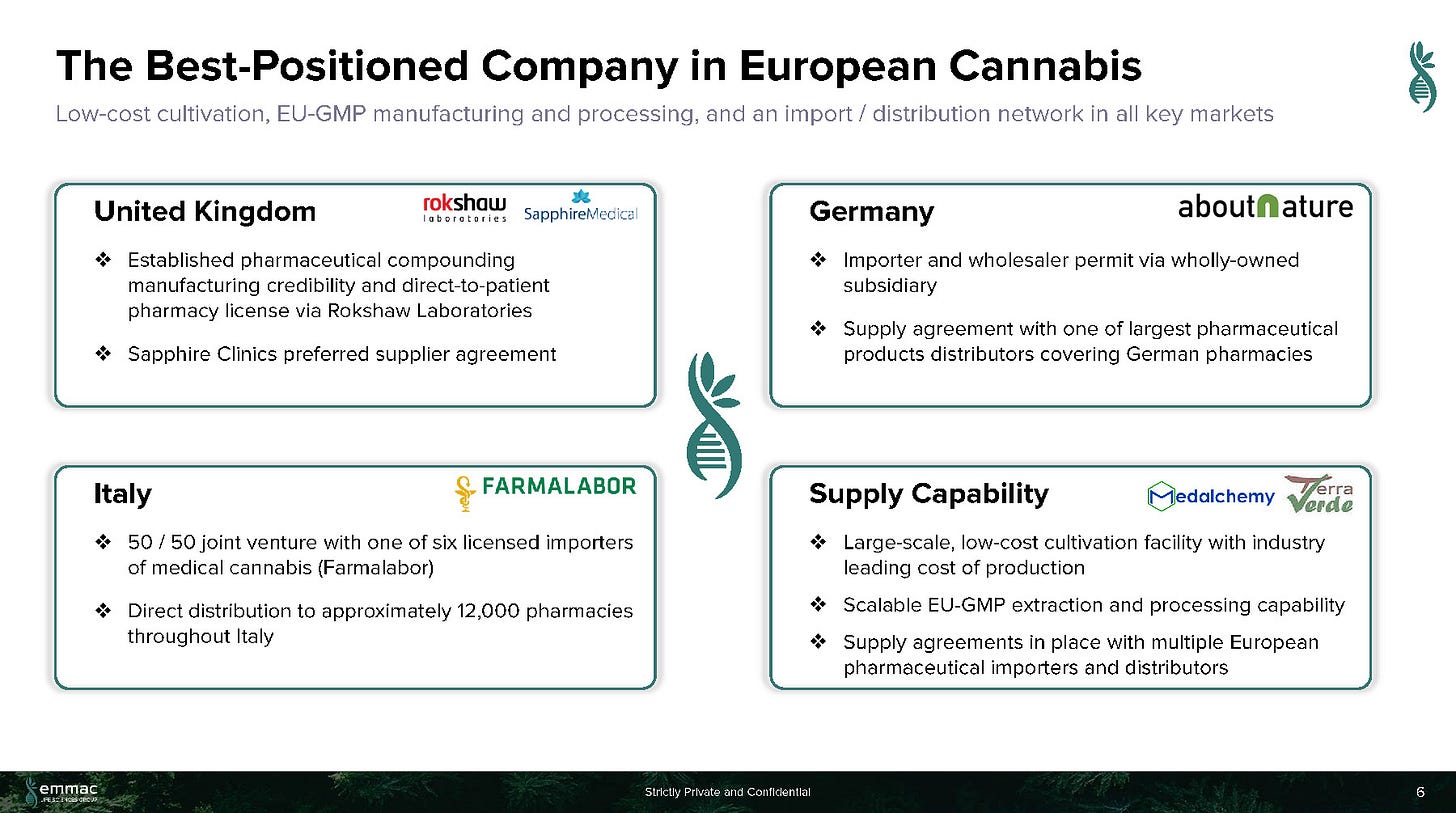
Footprint in Key European Counties (U.K., Germany, Italy, Switzerland & Portugal)
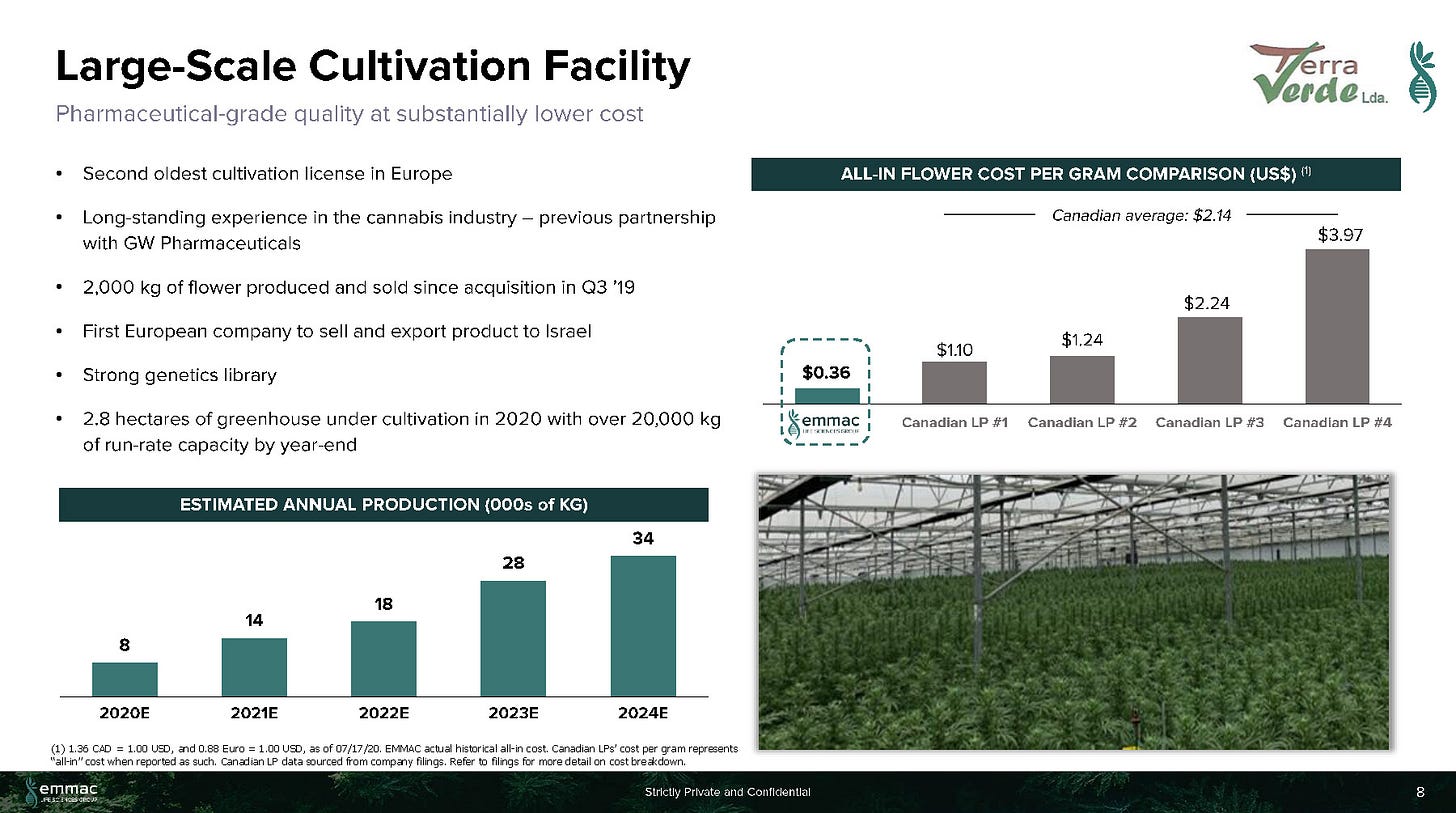
EMMAC's Portugal-based cultivation facility is an industry leader in cannabis flower production cost. Terra Verde, EMMAC's European market cultivation facility in Portugal is one of the oldest licensed cannabis growing facilities in Europe with ~2 hectares (~5 acres) of cultivation area. EMMAC plans to significantly increase its cultivation capacity in 2021, and to exceed 10 tons (20,000 lbs) per year by 2022.
Key Collaboration with Imperial College London
The initial focus (announced June 2019) was to research therapeutic products for chronic pain, spasticity and cancer.
EMMAC Timeline / Background:
Led by the former Head of International Development for Nuuvera, Antonio Costanzo, EMMAC seems to have grown via its experience dealing with government affairs, regulations and M&A. The Company had 130 employees (as of July 2020), with 17 PhDs given its focused on Medical Cannabis. The Company is vertically-integrated with Cultivation (20,000 lbs/year by 2022), Distribution (import/distribution in UK, Germany, Italy, Switzerland, Germany), Processing (2 operating EU-GMP facilities w/ another 2 under development), and R&D (pre-clinical research, collaborations with Imperial College and University Insubria). The Company had previously agreed to an LOI with a $100M SPAC (Andina Acquisition Corp. III) in July 2020, but the deal was terminated in November 2020.
March 2021 — Sells wellness brands for $8.1M to Yooma (CSE: YOOM), one of Asia’s leading CBD and wellness social commerce companies. Yooma will acquire the Blossom, MYO, Hello Joya and What the Hemp brands after previously licensing the brands from EMMAC under the terms of an agreement announced on January 20, 2021 for distribution and sale of the EMMAC Brands in China, Japan, Taiwan and Australia as well as non-exclusive distribution rights in certain other global markets.
January 2021 — Selected (in partnership with Paris Euronext listed Boiron) by the French Agency for the Safety of Medicines and Health Products (ANSM) as one of the suppliers for the forthcoming French Medical Cannabis trial. The trial, the first of its kind in France, will allow 3,000 patients access to medical cannabis as a treatment option for certain indications; including chronic pain, epilepsy, oncology and spasticity; under tightly controlled conditions. ANSM has selected 2 oral solutions produced by EMMAC as second-source treatments to be available to patients and healthcare professionals as part of the two year trial.
December 2020 — £15M raised via Convertible Notes, which saw strong support from existing shareholders, with Measure 8 Venture Partners, leading the round.
November 2020 — Andina Acquisition Corp. III (NASDAQ: ANDA) and EMMAC Life Sciences Limited (EMMAC) mutually terminated negotiations regarding the proposed business combination between the parties.
October 2020 — Rokshaw Laboratories, EMMAC’s wholly owned subsidiary and a leading UK MHRA-approved Specials Manufacturer, received its first commercial shipment of medical cannabis Active Pharmaceutical Ingredient (API) from Medalchemy, the Group’s Good Manufacturing Practice (GMP) certified manufacturing site in Alicante, Spain. Medalchemy will begin exporting medical cannabis API to other countries in the coming months.
August 2020 — EMMAC SAGL, the Group’s pharmaceutical entity in Switzerland, has secured its Good Distribution Practice (GDP) license for the distribution of medicinal products. EMMAC now has the ability to import, distribute and export medicinal products, including medical cannabis, for the domestic Swiss, EU and international markets.
July 2020 — Andina and EMMAC signed a non-binding LOI relating to a business combination with Andina Acquisition Corp. III (NASDAQ: ANDA). Medalchemy, the Group’s Good Manufacturing Practice (GMP) certified manufacturing site in Alicante, Spain has secured approval from the Spanish Health Authorities (AEMPS) to cultivate medical cannabis. Medalchemy now has fully integrated cannabis supply facilities, with a licence to grow medical cannabis commercially as well as extract and manufacture cannabis extract as an active pharmaceutical ingredient (API), which it received in May 2020.
March 2020 — About Nature, EMMAC’s wholly owned subsidiary, secured pharmaceutical wholesaler and narcotics handling permits for Germany. According to the BMO, the German medical cannabis market could be worth $5B+ in revenue for global cannabis producers, with an assumption that in seven years — 7.5% of sleep, anxiety and pain prescriptions in Germany will be replaced with medical cannabis. The U.K. also changed import restrictions to improve access to Cannabis-based medicines, allowing patients to get their treatment in days rather than months.
February 2020 — Launch of CBPM Access (a division of its wholly owned subsidiary, Rokshaw), the UK’s first operational distance pharmacy dedicated to fulfilling medical cannabis prescriptions. CBPM Access, as Rokshaw’s registered pharmacy, in conjunction with Rokshaw’s existing licenses, allows Rokshaw to import, manufacture and securely deliver medical cannabis products (CBPM) direct to UK-based patients. EMMAC also successfully exported 400 kilograms (~880 lbs) of medical cannabis to Israel, to the Bazelet Group, the largest medical cannabis company in Israel. The 400 kilograms of dried flower, the largest amount of product imported into Israel to date, was shipped from EMMAC’s cultivation facility, Terra Verde, in Portugal, making EMMAC the first independent European company to export medical cannabis to Israel.
November 2019 — Lorne Abony, former CEO of Nuuvera, joins as Chairman of the Board. Aphria acquired Nuuvera for $670M in January 2018 — the company’s footprint included Germany, Italy, Spain, UK, Malta, Israel, Lesotho and Uruguay.
October 2019 — £15M raised via Convertible Loan Notes. In addition to the initial seed capital of £2M, EMMAC raised a further £6M in January 2019, followed by £11M in March 2019 and with this latest CLN, £34M of funds has been raised by the Company in the last twelve months. The CLN Issue provides for notes to be converted to ordinary shares at a price of up to £0.50 per share giving EMMAC an implied value in excess of £150M.
July 2019 — acquired 100% of Terra Verde, a Portuguese genetics and cultivation company. Terra Verde holds oldest cannabis cultivation license in Portugal. The company also formed a 50/50 joint venture company, Fontana S.R.L. (Fontana JVCo), with Sergio Fontana, the founder and major shareholder of Farmalabor S.R.L., one of Italy’s most active licensed importers and distributors of medical cannabis. The partnership agreement with Farmalabor will provide Fontana JVCo with sales services, including distribution to 12,000 pharmacies. It is expected Fontana JVCo has the potential to rapidly establish itself as the largest supplier of medical cannabis and wellness products in Italy.
June 2019 — £11M raised, $2.4M from Altitude Investment Management, the firm’s thesis:
“While there is no real recreational market in Europe, the CBD wellness market is thriving, and the medical cannabis market is following a regulatory pathway that should ultimately lead to broad acceptance of doctor-prescribed cannabis and cannabinoid formulations.”
Acquired GreenLeaf, a French hemp-based and cannabis healthcare company.
April 2019 — Acquired UK-based Rockshaw Limited, which enabled EMMAC to import medical cannabis into the UK and to manufacture EU GMP products.
February 2019 — Rumored to be working with Canaccord Genuity on a listing that would have valued it at £400M. The company also formed a research partnership with UK biotechnology company Hyris Limited to develop a library of genetic profiles of existing cannabis varieties.